Classification of Antiretroviral Agents
Antiretroviral agents are synthetic antiviral medicines that have activity against human immunodeficiency virus (HIV) and are used in the management of HIV infection. There are currently five different classes of antiretroviral agents commercially available: nucleoside reverse transcriptase inhibitors (NRTIs), HIV protease inhibitors, non-nucleoside reverse transcriptase inhibitors (NNRTIs), nucleotide reverse transcriptase inhibitors, and HIV fusion inhibitors. These agents are often used in combination to suppress viral replication and reduce the risk of resistance.
| Class | Abbreviation | Representative agents | Primary target |
|---|---|---|---|
| Nucleoside reverse transcriptase inhibitors | NRTIs | Abacavir, zidovudine, lamivudine, stavudine | Reverse transcriptase (DNA chain termination) |
| HIV protease inhibitors | - | Indinavir, ritonavir, saquinavir, lopinavir | HIV protease (virion maturation) |
| Non-nucleoside reverse transcriptase inhibitors | NNRTIs | Delavirdine, efavirenz, nevirapine | Reverse transcriptase (allosteric inhibition) |
| Nucleotide reverse transcriptase inhibitors | - | Tenofovir disoproxil fumarate | Reverse transcriptase (DNA chain termination) |
| HIV fusion inhibitors | - | Enfuvirtide | Viral entry and fusion |
Nucleoside reverse transcriptase inhibitors
- abacavir sulfate (ABC)
- didanosine (ddI)
- emtricitabine (FTC)
- lamivudine (3TC)
- stavudine (dT)
- zalcitabine (ddC)
- zidovudine (ZDV, AZT)
NRTIs are synthetic analogues of naturally occurring nucleosides. There are currently seven NRTIs commercially available. These medicines have a wider spectrum of antiviral activity than other currently available antiretroviral agents, and NRTIs are active in vitro against human retroviruses, including HIV type 1 (HIV-1) and HIV type 2 (HIV-2). In addition, abacavir, didanosine, zalcitabine, and zidovudine are active against many animal retroviruses, including feline leukaemia virus, Friend leukaemia virus, Harvey murine sarcoma virus, murine leukaemia virus, and simian T-lymphotropic virus. Abacavir, didanosine, lamivudine, zalcitabine, and zidovudine have some activity against hepatitis B virus; however, NRTIs have generally been inactive against other human or animal viruses tested, including herpes simplex virus types 1 and 2, influenza virus, adenovirus, cytomegalovirus, respiratory syncytial virus, varicella-zoster virus, and vaccinia virus.
NRTIs are inactive until they are phosphorylated by cellular enzymes and converted into active triphosphate metabolites. The pharmacologically active triphosphate metabolites then compete with naturally occurring deoxynucleoside triphosphates for the active binding site on viral reverse transcriptase, an enzyme essential for viral replication. Once the triphosphate metabolites are incorporated into viral DNA, synthesis of the viral DNA chain is terminated because the metabolites lack a functional 3-hydroxyl group, which prevents further 5 to 3 phosphodiester linkages. This chain-termination effect is central to their antiviral activity.
HIV protease inhibitors
- amprenavir (VX-478)
- atazanavir sulfate (ATV)
- fosamprenavir calcium
- indinavir sulfate (IDV, MK-639)
- lopinavir and ritonavir (ABT-378/r)
- nelfinavir mesylate (AG1343)
- ritonavir (RTV, ABT-538)
- saquinavir (SQV, RO 31-8959)
HIV protease inhibitors are synthetic antiviral agents that were specifically designed based on the structure of HIV protease, an enzyme that plays an essential role in the HIV replication cycle. Computer models of the enzyme and its binding sites have helped to identify a wide variety of compounds as potential inhibitors of HIV protease activity. There are currently eight HIV protease inhibitors commercially available, and many more are under investigation.
HIV protease inhibitors have a very limited spectrum of activity. These medicines are active against HIV-1 and, to a lesser extent, some (but not all) are active against HIV-2. Indinavir and saquinavir have some activity against simian immunodeficiency virus; however, HIV protease inhibitors have generally been inactive against other human and animal viruses tested. On a molar basis, HIV protease inhibitors appear to be more active than either NRTIs or NNRTIs against susceptible HIV-1.
Currently available HIV protease inhibitors have similar mechanisms of action. These medicines inhibit replication of HIV-1 and HIV-2 by directly interfering with HIV protease activity. This results in the formation of non-infectious, immature virions. Most of these medicines act as selective, competitive, reversible inhibitors of the enzyme. Unlike NRTIs, the antiretroviral activity of most HIV protease inhibitors does not depend on intracellular conversion to an active metabolite. Because of their potency, these agents are key components of many combination regimens.
Non-nucleoside reverse transcriptase inhibitors
- delavirdine mesylate (DLV)
- efavirenz (EFV, DMP 266)
- nevirapine (NVP)

NNRTIs are a group of structurally diverse antiretroviral agents with a similar mechanism of action. There are currently three NNRTIs commercially available, and many more are under investigation.
NNRTIs have a very specific spectrum of activity. Currently available NNRTIs are active against HIV-1, but are inactive against HIV-2 and have also been inactive against all other human or animal viruses tested to date.
NNRTIs inhibit replication of HIV-1 by interfering with the viral RNA- and DNA-directed polymerase activities of reverse transcriptase. The mechanism of action of NNRTIs differs from that of NRTIs. While NRTIs interfere with reverse transcriptase activity by becoming incorporated into the growing viral DNA chain, NNRTIs interfere with reverse transcriptase function by binding directly to the enzyme in a non-competitive fashion. Unlike NRTIs, the antiretroviral activity of HIV NNRTIs does not depend on intracellular conversion to an active metabolite. This distinction has important implications for resistance patterns and combination therapy.
Nucleotide reverse transcriptase inhibitors
- tenofovir disoproxil fumarate

Nucleotide reverse transcriptase inhibitors are a group of acyclic nucleoside phosphonates with a similar mechanism of action. Tenofovir disoproxil fumarate is currently the only nucleotide reverse transcriptase inhibitor commercially available, but others are under investigation. For the purposes of therapeutic decisions, tenofovir is grouped with the NRTI class of antiretroviral agents.
Tenofovir disoproxil fumarate is inactive until it is hydrolysed in vivo to tenofovir and then phosphorylated by cellular enzymes and converted into tenofovir diphosphate. The diphosphate competes with naturally occurring deoxyadenosine triphosphate and, after incorporation into viral DNA, synthesis of the viral DNA chain is terminated. This mechanism closely parallels that of other reverse transcriptase chain terminators.
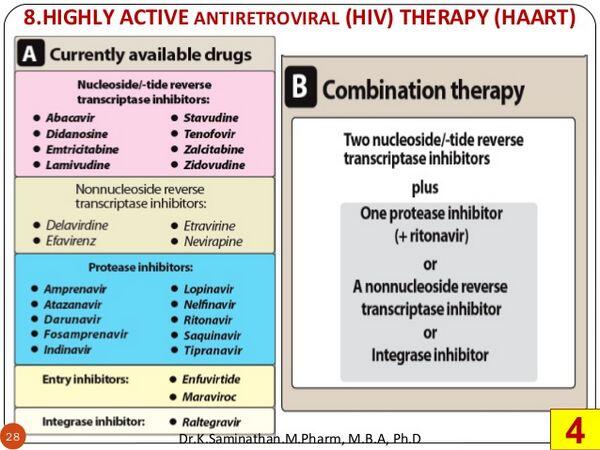
HIV fusion inhibitors
- enfuvirtide (T-20)

HIV fusion inhibitors are synthetic antiviral agents that interfere with the fusion of HIV-1 with its target cell. Enfuvirtide is currently the only HIV fusion inhibitor commercially available, but others are under investigation.
HIV fusion inhibitors have a very specific spectrum of activity. HIV fusion inhibitors are active against HIV-1, but are inactive against HIV-2.
HIV fusion inhibitors interfere with the entry of HIV-1 into target cells by blocking conformational changes in the HIV-1 glycoprotein that are required for fusion of the virus with the membrane of the host CD4+ T cell. By preventing this fusion step, these agents help to block the initial stages of viral infection of new cells.
| Class | Need for intracellular activation | Type of inhibition | Typical spectrum |
|---|---|---|---|
| NRTIs | Require phosphorylation to active triphosphate | Chain termination at reverse transcriptase | HIV-1 and HIV-2; some activity against hepatitis B virus |
| HIV protease inhibitors | No conversion to active metabolite required | Competitive inhibition of HIV protease | HIV-1; some agents active against HIV-2 |
| NNRTIs | No conversion to active metabolite required | Non-competitive binding to reverse transcriptase | HIV-1 only |
| Nucleotide reverse transcriptase inhibitors | Hydrolysis and phosphorylation to active diphosphate | Chain termination at reverse transcriptase | HIV-1 and HIV-2 |
| HIV fusion inhibitors | No conversion to active metabolite required | Block fusion and entry into CD4+ T cells | HIV-1 only |

















