Isoniazid

Dosages
Isoniazid 300 mg
| Quantity | Price per tablet | Total price | |
|---|---|---|---|
| 90 | £0.37 | £33.34 | |
| 120 | £0.34 | £40.74 | |
| 180 | £0.31 | £55.56 | |
| 270 | £0.29 | £78.52 | |
| 360 | £0.28 | £100.75 |
Payment & Delivery
Your order is carefully packed and is dispatched within 24 hours. Here is what a typical package looks like.
Sized like a regular personal letter (approximately 24x11x0.7 cm), with no indication of what is inside.



| Delivery Method | Estimated delivery |
|---|---|
| Express Free for orders over £222.24 | Estimated delivery to the UK: 4-7 days |
| Standard Free for orders over £148.16 | Estimated delivery to the UK: 14-21 days |










Discount Coupons
- New Year's Day - 1 January 2026 9% NEWYEAR9
- Valentine's Day - 14 February 2026 6% VALENTINE6
- St David's Day - 1 March 2026 4% DAVID4
- St Patrick's Day - 17 March 2026 5% PATRICK5
- St George's Day - 23 April 2026 4% GEORGE4
- May Day - 1 May 2026 5% MAYDAY5
- Halloween - 31 October 2026 6% HALLOWEEN6
- Guy Fawkes Night - 5 November 2026 5% GUY5
- Christmas Eve - 24 December 2026 9% XMASEVE9
- Christmas Day - 25 December 2026 10% XMAS10
- Boxing Day - 26 December 2026 10% BOXING10
- New Year's Eve - 31 December 2026 8% NYE8
Brand Names
| Country | Brand Names |
|---|---|
 Argentina Argentina | Isoniac |
 Austria Austria | INH Agepha |
 Belgium Belgium | Nicotibine Rimifon |
 Bulgaria Bulgaria | Isonid Rimicid |
 Canada Canada | Isotamine |
 Czechia Czechia | Nidrazid |
 Egypt Egypt | Isocid forte |
 Finland Finland | Tubilysin |
 France France | Rimifon |
 Germany Germany | Dipasic Gluronazid Isozid Isozid comp N Tb-Phlogin cum B6 tebesium tebesium-s |
 Greece Greece | Dianicotyl Isozid Nicozid |
 Hong Kong Hong Kong | Trisofort |
 Hungary Hungary | Isonicid |
 Iceland Iceland | Tibinide |
 India India | Isokin Isonex Rifacom E-Z |
 Israel Israel | Inazid |
 Italy Italy | Cin Nicazide Nicizina Nicozid |
 Japan Japan | Hydra Hydrazide |
 Mexico Mexico | Dipasic Erbazid Hidrasix Pas Hain Valifol |
 Pakistan Pakistan | Aceta Amsozide Isonex Forte |
 Philippines Philippines | Isonid |
 Portugal Portugal | Hidrazida |
 Romania Romania | Bitub |
 Slovenia Slovenia | INH Agepha |
 Spain Spain | Anidrasona Cemidon Cemidon B6 Dipasic Hidrastol Pyreazid Rimifon |
 Sweden Sweden | Tibinide |
 Switzerland Switzerland | Rimifon |
 Taiwan Taiwan | Duracrin I.N.A.H. Iscotin |
 Turkey Turkey | I.N.H. |
 United Kingdom United Kingdom | Inapsade Rimifon |
 United States United States | Laniazid Nydrazid |
Description
Isoniazid is a first-line medicine for tuberculosis. It is used to treat active tuberculosis (together with other medicines) and to help prevent active disease in people with latent tuberculosis (TB) infection. In the UK, it should only be used under appropriate clinical supervision, as correct dosing and monitoring are important, especially for liver safety. Follow your clinician's instructions carefully.

Uses
Active Tuberculosis
Isoniazid is used with other antituberculosis medicines to treat clinical tuberculosis.
In the UK, treatment of culture-positive pulmonary tuberculosis generally follows NICE guidance and specialist TB protocols, with several recommended multidrug regimens. These regimens last for at least 6 months (26 weeks) and include an initial intensive phase (2 months) followed by a continuation phase (usually 4 or 7 months). Isoniazid is considered a first-line antituberculosis medicine for treating all forms of tuberculosis caused by Mycobacterium tuberculosis known or thought to be susceptible to the drug.
In the UK, isoniazid is available on its own and in fixed-dose combinations used to treat tuberculosis, including combinations with rifampicin and with rifampicin plus pyrazinamide where licensed products are available. Although oral isoniazid is preferred for treating tuberculosis, the drug may be given IM for initial treatment or retreatment when it cannot be taken by mouth.
Latent Tuberculosis Infection
Isoniazid is usually used on its own to treat latent tuberculosis infection and help prevent clinical tuberculosis from developing. In the past, terms such as "preventive therapy" or "chemoprophylaxis" were used for a simple drug regimen, such as isoniazid on its own, given to prevent active tuberculosis in people known or likely to be infected with M. tuberculosis. However, in UK clinical practice, "treatment of latent tuberculosis infection" is generally the preferred term because it more accurately describes the intended treatment and supports wider understanding and use of this tuberculosis control strategy.
People at risk of developing tuberculosis include those who have recently been infected with M. tuberculosis and those who have clinical conditions that increase the risk of latent tuberculosis infection progressing to active disease. The chance that a positive tuberculin test reflects a true infection with M. tuberculosis depends partly on how common the infection is in the population being tested. In the UK, tuberculin testing and other tests for latent tuberculosis are generally aimed at groups at increased risk, rather than used widely in people at low risk, to improve the predictive value of testing and reduce false-positive results. Interpretation of tuberculin skin test results is based on the size of the induration together with the patient's risk factors, clinical context, and local guidance.
Key Groups for Tuberculin Testing and Treatment
- Individuals with HIV Infection
- Those with an induration reaction of 5 mm or more should receive LTBI therapy unless it is contraindicated.
- Preventive therapy is recommended even with a negative tuberculin test if there is known exposure to active tuberculosis.
- Isoniazid therapy may be beneficial for tuberculin-negative children born to mothers with HIV infection.
- Close Contacts of Tuberculosis Patients
- Contacts with a significant reaction (≥5 mm) should be treated for LTBI, regardless of age.
- Children under 5 years should be treated regardless of test results because they are more susceptible to the disease.
- Immunocompromised Individuals
- Those receiving prolonged corticosteroid therapy or organ transplants should be treated if they have a significant tuberculin reaction.
- Immunosuppressed individuals who are contacts of active tuberculosis cases should also receive treatment.
- Individuals with a Prior Tuberculosis History
- Those with healed but untreated tuberculosis should receive LTBI therapy, regardless of age.
- High-Risk Population Groups
- Recent immigrants from high-prevalence countries, residents in long-term care facilities, and healthcare staff exposed to tuberculosis patients should be considered for treatment if they have significant tuberculin reactions.
- Children and Adolescents
- Infants and children exposed to high-risk adults should be treated if they have a significant tuberculin reaction.
Testing and Treatment Considerations
- Routine testing is not recommended for low-risk populations, but treatment may be considered for those with significant reactions.
- Before starting isoniazid therapy, patients must be checked for active tuberculosis and liver conditions to make sure there are no contraindications.
This approach helps ensure that vulnerable groups receive appropriate screening and treatment to reduce the risk of latent tuberculosis progressing to active disease.
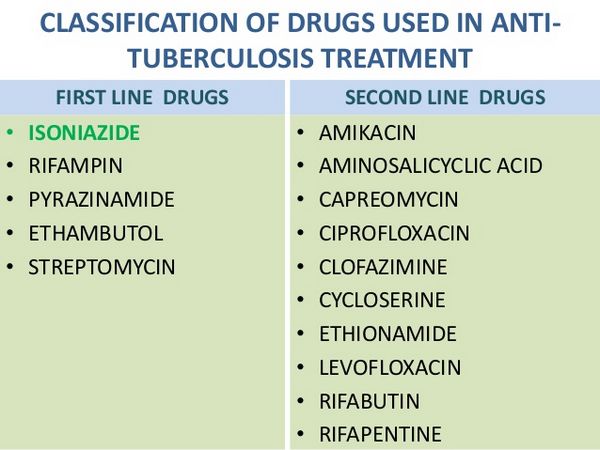
Isoniazid Monotherapy
Isoniazid monotherapy is recommended for treating latent tuberculosis infection (LTBI) in both adults with HIV infection and those without HIV, with a preferred regimen of 9 months taken daily or twice weekly. For infants and children, a similar 9-month regimen is advised, although some experts suggest extending treatment to 12 months for children with HIV infection. While the 9-month regimen is preferred, a 6-month regimen can be used for adults without HIV, offering substantial protection and potentially lower costs. However, it is not recommended for children or for those with prior evidence of tuberculosis. Research suggests that regimens shorter than 6 months are ineffective, and a large study confirmed that a 6-month course is more effective than shorter options. UK practice also places strong emphasis on adherence, recommending completion according to the total number of prescribed doses rather than duration alone. Intermittent dosing should be directly observed to support adherence. If treatment is interrupted for more than two months, a medical review is needed before restarting therapy.
Alternative Regimens
While isoniazid monotherapy is generally the treatment of choice for latent tuberculosis infection, a 4-month course of daily rifampin monotherapy can be used as an alternative in both HIV-positive and HIV-negative patients, especially when isoniazid cannot be used because of resistance or intolerance.
Limited data suggest that a short-course (for example, 2-month) regimen of daily rifampin and pyrazinamide is effective for treating latent tuberculosis infection in patients with HIV, and similar efficacy would not be expected to differ in HIV-negative patients. However, hepatotoxicity, including some fatal cases, has been reported in patients receiving rifampin and pyrazinamide regimens for latent tuberculosis. Although multidrug regimens containing rifampin and pyrazinamide are still recommended for active tuberculosis, current UK practice is that regimens containing both rifampin and pyrazinamide generally should not be offered for latent tuberculosis infection in either HIV-positive or HIV-negative individuals.
HIV-infected Individuals
Factors to consider when choosing the most appropriate regimen for latent tuberculosis infection in people with HIV include how likely it is that the infecting organism is susceptible to isoniazid, as isoniazid is the preferred treatment for isoniazid-susceptible M. tuberculosis, the potential for drug interactions with rifampin in patients taking HIV protease inhibitors or non-nucleoside reverse transcriptase inhibitors (NNRTIs), and the possibility of severe liver injury with pyrazinamide-containing regimens. If the infecting organism is resistant to both isoniazid and rifampin, the choice of treatment requires consultation with public health authorities.
Recommendations for treating latent tuberculosis infection in adults with HIV are generally similar to those for HIV-negative adults. However, the 6-month isoniazid monotherapy regimen is usually not recommended, and rifabutin monotherapy may be needed instead of rifampin monotherapy if there are concerns about interactions with antiretroviral medicines the patient may be taking. In UK practice, adults and adolescents with HIV and latent M. tuberculosis infection may receive a 9-month regimen of isoniazid given daily or twice weekly, or a 4-month regimen of rifampin or rifabutin given daily. A 2- to 3-month regimen of rifampin and pyrazinamide is no longer recommended for most patients.
For infants and children with HIV, recommended regimens for latent tuberculosis infection are isoniazid given daily or twice weekly for 9 to 12 months, or rifampin given daily for 4 to 6 months.
Pregnant Women
For pregnant women at risk of latent tuberculosis infection progressing to active disease, particularly those with HIV infection or recent infection, UK guidance indicates that starting or stopping treatment for latent tuberculosis infection should not be delayed because of pregnancy alone, even during the first trimester. For women whose risk of active disease is lower, some experts recommend delaying treatment until after delivery. Patients with HIV infection or radiographic evidence of prior tuberculosis should receive 9 rather than 6 months of isoniazid therapy. In pregnant women without HIV infection, a regimen of isoniazid given daily or twice weekly for 9 or 6 months is recommended.
Drug-Resistant Latent Tuberculosis Infection
In individuals likely to be infected with M. tuberculosis organisms resistant to both isoniazid and rifampin and who are at high risk of developing tuberculosis, specialist regimens such as pyrazinamide and ethambutol, or pyrazinamide and a quinolone anti-infective (for example, levofloxacin or ofloxacin) for 6-12 months may be considered if the organisms from the index case are known to be susceptible to these drugs. Immunocompetent contacts may be managed with observation alone or treated with such regimens for 6 months; immunosuppressed individuals, including those with HIV infection, should be treated for 12 months. Clinicians should review the drug-susceptibility pattern of the M. tuberculosis strain isolated from the source patient before choosing a regimen for potentially multidrug-resistant tuberculosis infections. In individuals likely to have been infected with M. tuberculosis organisms resistant to both isoniazid and rifampin, the choice of medicines for latent infection requires expert advice. Before starting treatment for latent tuberculosis infection in patients with suspected multidrug-resistant tuberculosis, careful assessment is needed to rule out active disease.
In UK paediatric practice, until susceptibility test results are available, contacts likely to have been infected by an index case with isoniazid-resistant tuberculosis may receive both rifampin and isoniazid. If the index case is proven to be excreting organisms that are completely resistant to isoniazid, isoniazid should be stopped and rifampin should be given for at least six months. Expert advice is recommended when deciding on therapy for latent tuberculosis infection in children with isoniazid- and/or rifampin-resistant M. tuberculosis.
Dosage
Oral and IM doses of isoniazid are identical.
Active Tuberculosis
In the treatment of clinical tuberculosis, isoniazid should not be given alone. The drug is considered a first-line treatment for all forms of tuberculosis. Treatment should continue for long enough to help prevent relapse. In the UK, the minimum recommended treatment duration for patients with culture-positive pulmonary tuberculosis is generally 6 months (26 weeks), with an initial intensive phase of 2 months followed by a continuation phase of usually 4 or 7 months. In practice, completion of treatment is assessed more accurately by the total number of doses received and the overall regimen completed, rather than by duration alone.
Adult Dosage
When isoniazid is used with other antituberculosis medicines, UK guidance usually recommends 300 mg once daily for adults. For children, the usual dose is 10 mg/kg/day, up to a maximum of 300 mg per dose.
Pediatric Dosage
Infants and children tolerate larger doses of isoniazid than adults and may be given isoniazid in a dose of up to 10-20 mg/kg/day, depending on the severity of the disease. The maximum dose of isoniazid recommended by the manufacturers for children is 300-500 mg daily. In UK paediatric tuberculosis practice, when isoniazid is used in daily multidrug regimens in children, a dose of 10-15 mg/kg/day (up to 300 mg) is generally used. Caution is advised because using an isoniazid dose above 10 mg/kg/day together with rifampin may increase the incidence of hepatotoxicity.
When an intermittent multidrug regimen is used to treat tuberculosis in children, UK guidance recommends isoniazid 15 mg/kg three times weekly, up to a maximum of 900 mg per dose. Daily supervised treatment is preferred where feasible.
Fixed-Combination Preparations
When isoniazid is given as the fixed combination containing isoniazid and rifampin (Rifamate®) as part of a multidrug regimen for pulmonary tuberculosis, the usual adult dose of Rifamate® is 2 capsules (600 mg of rifampin and 300 mg of isoniazid) once daily.
Although the fixed-combination preparation was formulated for daily regimens, UK tuberculosis practice allows Rifamate® to be used in twice-weekly regimens provided additional isoniazid is given at the same time.
When used in an intermittent multidrug regimen, these experts state that two capsules of Rifamate® (600 mg of rifampin and 300 mg of isoniazid) and an additional 600 mg of isoniazid (900 mg of isoniazid in total) may be given twice weekly using directly observed therapy (DOT).
The manufacturer states that Rifamate® should not be used for the initial treatment of tuberculosis, but only after the effectiveness of the rifampin and isoniazid doses contained in the fixed-combination preparation has been established by titrating the individual components in the patient.
When isoniazid is given as the fixed combination containing isoniazid, rifampin, and pyrazinamide (Rifater®) in the initial phase (for example, the first 2 months) of multidrug therapy for pulmonary tuberculosis, the manufacturer states that the adult dose of Rifater® given as a single daily dose is 4 tablets (480 mg of rifampin, 200 mg of isoniazid, 1.2 g of pyrazinamide) in patients weighing 44 kg or less, 5 tablets (600 mg of rifampin, 250 mg of isoniazid, and 1.5 g of pyrazinamide) in those weighing 45-54 kg, and 6 tablets (720 mg of rifampin, 300 mg of isoniazid, 1.8 g of pyrazinamide) in patients weighing 55 kg or more. In individuals weighing more than 90 kg, additional pyrazinamide may need to be given with the fixed-combination preparation to obtain an adequate dose of this drug.
The ratio of rifampin, isoniazid, and pyrazinamide in Rifater® may not be appropriate in children or adolescents under 15 years of age because children usually receive higher mg/kg doses of isoniazid than adults.
Latent Tuberculosis Infection
Isoniazid is usually the only antituberculosis medicine used for a minimum of six months to treat latent tuberculosis infection. Every effort should be made to support adherence for at least six months, since preventive therapy of shorter duration appears to provide little benefit. If the medicine cannot be given under direct observation, spot testing of urine for isoniazid metabolites has been recommended to assess adherence.
In the UK, a 9-month daily isoniazid regimen or, alternatively, a 9-month twice-weekly isoniazid regimen may be used for adults regardless of HIV status. Continuing isoniazid therapy for latent tuberculosis infection for longer than 12 months provides no additional benefit. It is also recommended that isoniazid therapy for latent tuberculosis infection be continued for 9-12 months in infants and children with HIV.
In UK practice, completion of therapy for latent tuberculosis infection is determined more accurately by the total number of doses and should not be based on duration alone. The 9-month daily isoniazid regimen should include at least 270 doses given within 12 months, allowing for interruptions in the usual 9-month regimen, and the 6-month daily isoniazid regimen should include at least 180 doses given within 9 months. Isoniazid regimens in which the drug is given twice weekly should include at least 76 doses within 12 months for the 9-month regimen or at least 52 doses within 9 months for the 6-month regimen.
Administration
Isoniazid is usually taken by mouth. The drug may be given by IM injection when oral treatment is not possible. The fixed-combination preparation containing isoniazid and rifampin (Rifamate®) and the fixed-combination preparation containing isoniazid, rifampin, and pyrazinamide (Rifater®) should be taken either 1 hour before or 2 hours after a meal; the manufacturer states that Rifater® should be taken with a full glass of water.
Solutions of isoniazid should be sterilised by autoclaving.
Important Safety Information
Liver function tests should be carried out regularly in patients taking isoniazid. Patients should also be asked each month about signs and symptoms of liver disease and told to report any early symptoms of hepatitis to their doctor (for example, persistent tiredness, weakness, or a fever lasting more than 3 days; feeling unwell; nausea; vomiting; unexplained loss of appetite). If these symptoms appear, or if signs suggesting liver damage occur, isoniazid should be stopped promptly, since continuing treatment in these patients has been reported to cause a more severe form of liver injury.
Some clinicians recommend stopping isoniazid if serum aminotransferase levels are more than 3-5 times the upper limit of normal, or if the patient develops signs of hepatitis. Patients who have had signs or symptoms of liver damage during isoniazid treatment should generally be given alternative anti-tuberculosis medicines, but if isoniazid has to be started again, it should only be restarted after liver symptoms and laboratory abnormalities have resolved. Isoniazid should be restarted at very small doses, increased gradually, and stopped immediately if there is any sign of liver problems returning.
UK paediatric guidance says that hepatitis during isoniazid treatment in children is rare and that routine measurement of serum aminotransferase levels is not usually recommended. However, liver function tests should be monitored about monthly during the first few months of treatment in children with severe tuberculosis, especially meningitis and disseminated disease.
UK paediatric guidance also advises monitoring liver function tests in patients with current or recent liver disease, those receiving a high daily dose of isoniazid (more than 10 mg/kg/day) together with rifampicin and/or pyrazinamide, those who are pregnant or within 6 weeks after giving birth, those with clinical signs of hepatotoxicity, those with hepatobiliary disease from other causes, and those taking other hepatotoxic drugs at the same time (especially anticonvulsants). In most other patients, monthly clinical review for 3 months, followed by review every 1-3 months to check for signs of hepatitis or other side effects of treatment, is appropriate.
Isoniazid should be used with caution in people who drink alcohol daily, people who inject illicit drugs, patients with chronic liver disease or severe renal impairment, and those with a history of previous treatment in whom isoniazid was stopped because of side effects (for example, headache, dizziness, nausea) that were possibly, but not definitely, related to the drug. Small dose adjustments may be needed in patients with severe renal impairment. Limited data from a retrospective analysis of deaths from isoniazid-associated hepatitis suggest that the risk of fatal hepatitis with the drug may be higher in women, particularly Black and Hispanic women, and during the postpartum period.
Regular ophthalmic examinations should be carried out in patients who develop visual symptoms while taking the drug. The manufacturers recommend ophthalmic examinations (including ophthalmoscopy) before starting isoniazid and regularly during treatment, even if visual symptoms do not occur; however, some clinicians question whether this precaution is necessary.
Isoniazid should be used with caution in patients who are malnourished or prone to neuropathy (for example, people with diabetes or alcohol use disorder), and pyridoxine should generally be given at the same time. In UK paediatric practice, pyridoxine is recommended in children and adolescents with an abnormally low intake of milk and meat, in those with nutritional deficiencies (including all children with symptomatic HIV infection), in breast-fed infants and their mothers, and in pregnant women.
Isoniazid is contraindicated in patients with acute liver disease or a history of previous isoniazid-associated liver injury. Although treatment for latent tuberculosis infection should be deferred in these patients, UK guidance indicates that seropositivity for hepatitis B surface antigen is not, by itself, a contraindication to this treatment. Isoniazid is also contraindicated in patients with a history of severe adverse reactions to the drug, including severe hypersensitivity reactions or drug fever, chills, and arthritis.
Side effects
Isoniazid is generally well tolerated at the currently recommended doses. However, people who are slow acetylators of isoniazid and those with advanced HIV disease appear to have a higher rate of some side effects. Patients with poor nutrition are also at risk of peripheral neuritis, which is one of the most common side effects of isoniazid. Other neurological side effects include psychotic reactions and convulsions. Pyridoxine may be given to prevent or treat these side effects. Optic neuritis has also been reported. Temporary increases in liver enzymes occur in 10 to 20% of patients during the first few months of treatment and usually return to normal despite continued treatment. Symptomatic hepatitis occurs in about 0.1 to 0.15% of patients given isoniazid on its own, but this can increase with age, regular alcohol use, and chronic liver disease. The effect of acetylator status is uncertain.
Raised liver enzymes together with clinical signs of hepatitis, such as nausea, vomiting, or fatigue, may indicate liver damage. In these circumstances, isoniazid should be stopped pending assessment and should only be restarted cautiously once liver function has recovered. Fatalities have occurred due to liver necrosis.
Interactions
The risk of hepatotoxicity may be increased in patients taking isoniazid with a rifamycin or other potentially hepatotoxic drugs, including alcohol. Isoniazid can inhibit the liver metabolism of a number of drugs, in some cases leading to increased toxicity. These include the antiepileptics carbamazepine, ethosuximide, primidone, and phenytoin, the benzodiazepines diazepam and triazolam, chlorzoxazone, theophylline, and disulfiram. Isoniazid has been associated with increased concentrations and enhanced effects or toxicity of clofazimine, cicloserine, and warfarin.
Mechanism of action
Isoniazid may be bacteriostatic or bactericidal, depending on the concentration of the drug reached at the site of infection and the susceptibility of the infecting organism.
Although the exact way isoniazid works has not been fully clarified, several mechanisms have been proposed, including interference with the metabolism of bacterial proteins, nucleic acids, carbohydrates, and lipids.
One of the drug's main actions appears to be inhibition of mycolic acid synthesis in susceptible bacteria, resulting in loss of acid-fastness and disruption of the bacterial cell wall. Isoniazid is active against susceptible bacteria only when they are undergoing cell division. Susceptible bacteria may undergo 1 or 2 divisions before multiplication is arrested. Isoniazid is a highly specific agent and is active only against organisms of the genus Mycobacterium. Isoniazid is active in vitro and in vivo against M. tuberculosis, M. bovis, and some strains of M. kansasii.
Resistance
Natural and acquired resistance to isoniazid has been demonstrated in vitro and in vivo in strains of M. tuberculosis. In vitro, resistance to isoniazid develops step by step. The mechanism of resistance may be related to the drug's failure to penetrate or be taken up by resistant bacteria.
Resistant strains of bacteria that were initially susceptible develop rapidly if isoniazid is used alone to treat clinical tuberculosis. However, the development of resistance does not appear to be a major problem when the drug is used alone for preventive therapy. When isoniazid is combined with other anti-tuberculosis agents in the treatment of clinical tuberculosis, the emergence of resistant strains may be delayed or prevented.
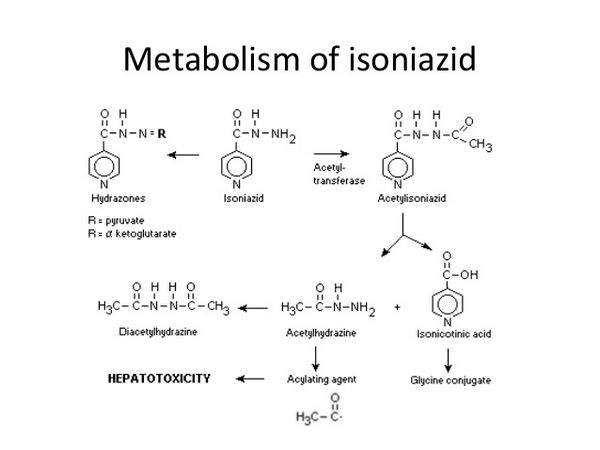
Pharmacokinetics
Isoniazid is readily absorbed from the gastrointestinal tract and after intramuscular injection. Peak concentrations of about 3 to 7 micrograms/mL appear in the blood 1 to 2 hours after an oral fasting dose of 300 mg. Food reduces the rate and extent of isoniazid absorption. Isoniazid is not thought to bind significantly to plasma proteins and is distributed into all body tissues and fluids, including the cerebrospinal fluid (CSF). It appears in fetal blood if given during pregnancy and is distributed into breast milk. The plasma half-life of isoniazid ranges from about 1 to 6 hours, with shorter half-lives in fast acetylators. The main metabolic pathway is acetylation of isoniazid to acetyl isoniazid by N-acetyltransferase in the liver and small intestine. In patients with normal renal function, more than 75% of a dose appears in the urine within 24 hours, mainly as metabolites. Small amounts of the drug are also excreted in the faeces. Isoniazid is removed by haemodialysis.
Distribution
Therapeutic concentrations of isoniazid have been detected in CSF and synovial fluid several hours after an oral dose. It diffuses well into saliva, and it has been suggested that salivary concentrations could be used instead of serum concentrations in pharmacokinetic studies.
HIV-infected patients
Malabsorption of isoniazid and other anti-tuberculosis drugs may occur in patients with HIV infection and tuberculosis and may contribute to acquired drug resistance and reduced effectiveness of tuberculosis treatment.
Pregnancy
Isoniazid crosses the placenta, and average fetal concentrations of 61.5 and 72.8% of maternal serum or plasma concentrations have been reported. The half-life of isoniazid may be prolonged in neonates.
Storage
Isoniazid preparations should be protected from light, air, and excessive heat. Isoniazid tablets should be stored in tightly closed, light-resistant containers at a temperature below 40°C, preferably between 15-30°C. Tablets containing the fixed combination of rifampicin, isoniazid, and pyrazinamide (Rifater®) should be protected from excessive humidity and stored at 15-30°C. Isoniazid injection should be protected from light and stored at a temperature below 40°C, preferably between 15-30°C; freezing should be avoided. At low temperatures, isoniazid in solution tends to crystallise, and the injection should be warmed to room temperature to redissolve the crystals before use.
Overdosage
Too much isoniazid can cause nausea, vomiting, dizziness, slurred speech, blurred vision, and visual hallucinations (including bright colours and strange patterns). Symptoms of overdose usually occur within 30 minutes to 3 hours after taking the drug. After marked overdose, respiratory distress and CNS depression, progressing rapidly from stupor to coma, severe intractable seizures, metabolic acidosis, acetonuria, and hyperglycaemia have occurred. If untreated or treated inadequately, isoniazid overdose may be fatal.
Treatment of overdose
When managing isoniazid overdose, the airway should be secured and adequate breathing established immediately. Seizures may be controlled with IV diazepam or short-acting barbiturates and a dose of pyridoxine hydrochloride equal to the amount of isoniazid ingested. Generally, 1-4 g of pyridoxine hydrochloride is given IV, followed by 1 g IM every 30 minutes until the full dose has been given. If seizures are controlled and the overdose is recent (within 2-3 hours), the stomach should be emptied by gastric lavage.
Blood gases, serum electrolytes, glucose, and blood urea nitrogen should be measured. Blood should be typed and cross-matched in case haemodialysis is needed. Intravenous sodium bicarbonate should be given to control metabolic acidosis and repeated if necessary. The dose should be adjusted according to laboratory test results.